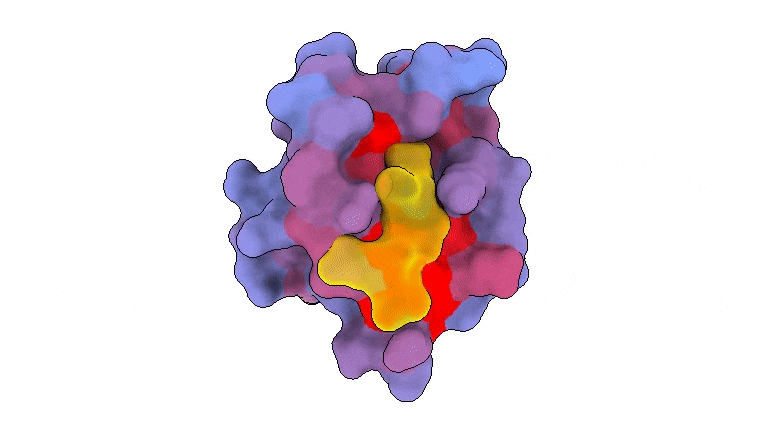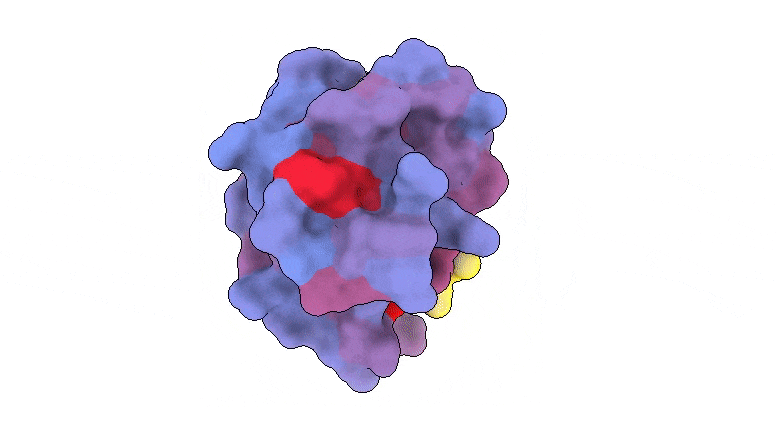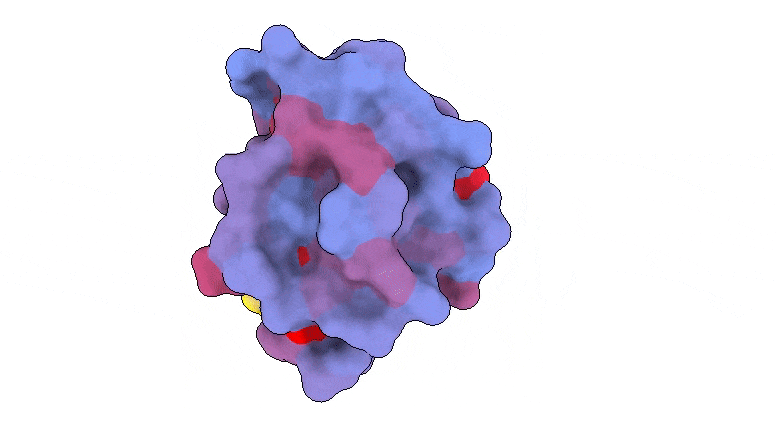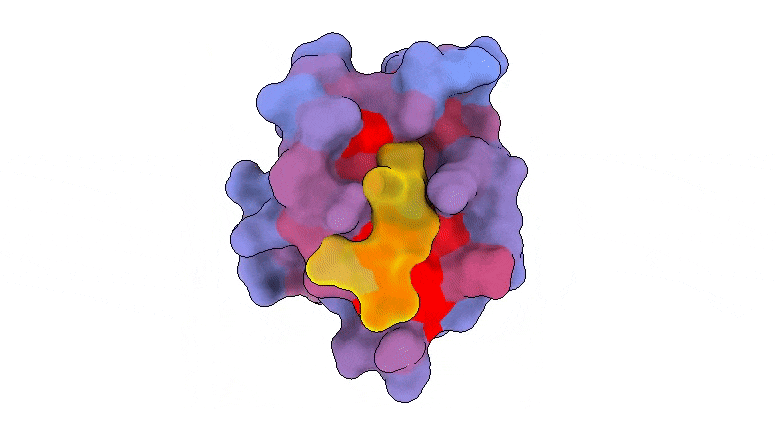
A 3-dimensional animation of the human protein PSD95-PDZ3 displaying the binding accomplice CRIPT (yellow) within the lively web site with the blue-to-red colour gradient indicating growing potential for allosteric results. Primarily based on PDB accession 1BE9. Credit score: André Faure/CRG
Identification of hidden vulnerabilities on floor of ‘undruggable’ proteins may remodel therapy of illness.
The variety of potential therapeutic targets on the surfaces of human proteins is far larger than beforehand thought, in response to the findings of a brand new examine within the journal Nature.
A ground-breaking new method developed by researchers on the Heart for Genomic Regulation (CRG) in Barcelona has revealed the existence of a mess of beforehand secret doorways that management protein perform and which may, in principle, be focused to dramatically change the course of situations as diversified as dementia, most cancers and infectious ailments.
The tactic, wherein tens of hundreds of experiments are carried out on the identical time, has been used to chart the primary ever map of those elusive targets, also referred to as allosteric websites, in two of the commonest human proteins, revealing they're ample and identifiable.
The method may very well be a game-changer for drug discovery, resulting in safer, smarter and more practical medicines. It allows analysis labs all over the world to seek out and exploit vulnerabilities in any protein – together with these beforehand thought ‘undruggable’.
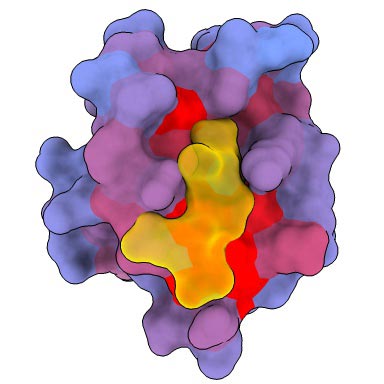
A 3-dimensional picture displaying the human protein PSD95-PDZ3 from the entrance. Proven is the binding accomplice CRIPT (yellow) within the lively web site with the blue-to-red colour gradient indicating growing potential for allosteric results. Primarily based on PDB accession 1BE9. Credit score: André Faure/CRG
“Not solely are these potential therapeutic websites ample, there may be proof they are often manipulated in many alternative methods. Somewhat than merely switching them on or off, we may modulate their exercise like a thermostat. From an engineering perspective, that’s hanging gold as a result of it offers us loads of area to design ‘good medication’ that focus on the unhealthy and spare the nice,” explains André Faure, postdoctoral researcher on the CRG and co-first writer of the paper.
Proteins play a central function in all dwelling organisms and perform important capabilities corresponding to offering construction, rushing up reactions, performing as messengers or preventing illness. They're manufactured from <div class="text-wrapper"><br />Amino acids are a set of organic compounds used to build proteins. There are about 500 naturally occurring known amino acids, though only 20 appear in the genetic code. Proteins consist of one or more chains of amino acids called polypeptides. The sequence of the amino acid chain causes the polypeptide to fold into a shape that is biologically active. The amino acid sequences of proteins are encoded in the genes. Nine proteinogenic amino acids are called "essential" for humans because they cannot be produced from other compounds by the human body and so must be taken in as food.<br /></div>"
</div></div>' data-gt-translate-attributes='["attribute":"data-cmtooltip", "format":"html"]'>amino acids, folding into numerous completely different shapes in three-dimensional area. The form of a protein is essential for its perform, with only one mistake in an amino acid sequence leading to doubtlessly devastating penalties for human well being.
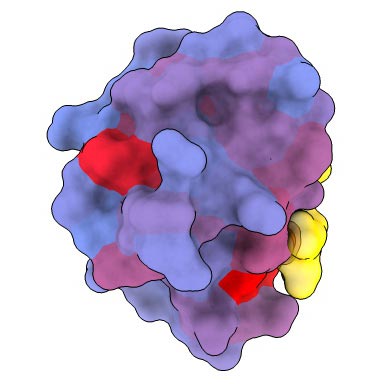
A 3-dimensional picture displaying the human protein PSD95-PDZ3 from the again. Proven is the binding accomplice CRIPT (yellow) within the lively web site with the blue-to-red colour gradient indicating growing potential for allosteric results. Primarily based on PDB accession 1BE9. Credit score: André Faure/CRG
Allostery is likely one of the nice unsolved mysteries of protein perform. Allosteric results happen when a molecule binds to the floor of a protein, which in flip causes modifications at a distant web site in the identical protein, regulating its perform by distant management. Many disease-causing mutations, together with quite a few most cancers drivers, are pathological due to their allosteric results.
Regardless of their elementary significance, allosteric websites are extremely tough to seek out. It's because the foundations governing how proteins work on the atomic stage are hidden out of sight. For instance, a protein may shapeshift within the presence of an incoming molecule, revealing hidden pockets deep inside its floor which can be doubtlessly allosteric however not identifiable utilizing standard construction dedication alone.
Drug hunters have historically designed therapies that focus on a protein’s lively web site, the small area the place chemical reactions happen or targets are sure. The draw back of those medication, also referred to as orthosteric medication, is that lively websites of many proteins look very comparable and so medication are likely to bind and inhibit many alternative proteins directly, resulting in potential unintended effects. Compared, the specificity of allosteric websites implies that allosteric medication are a few of the handiest kinds of treatment at present obtainable. Many allosteric medication, which deal with numerous situations starting from most cancers to AIDS to hormone problems, have been found accidentally.
The authors of the examine addressed this problem by creating a way referred to as double deep PCA (ddPCA), which they describe as a ‘brute power experiment’. “We purposefully break issues in hundreds of various methods to construct an entire image of how one thing works,” explains ICREA Analysis Professor Ben Lehner, Coordinator of the Methods Biology program on the CRG and writer of the examine. “It’s like suspecting a defective spark plug, however as an alternative of solely checking that, the mechanic dismantles your entire automotive and checks it piece by piece. By testing ten thousand issues in a single go we establish all of the items that basically matter.”
The tactic works by altering the amino acids that make up a protein, leading to hundreds of various variations of the protein with only one or two variations within the sequence. The consequences of the mutations are then examined all on the identical time in dwelling cells within the laboratory.
“Every cell is a tiny manufacturing unit making a unique model of the protein. In a single check tube we now have tens of millions of various factories and so we will very quickly check how properly all of the completely different variations of a protein work,” provides Dr. Lehner. The info collected from the experiments is fed into neural networks, algorithms that analyze knowledge by mimicking the best way the human mind operates, which end in complete maps that pinpoint the situation of allosteric websites on the surfaces of proteins.
One of many nice benefits of the tactic is that it's an reasonably priced method accessible to any analysis lab all over the world. “It massively simplifies the method wanted to seek out allosteric websites, with the method working at a stage of accuracy higher than a number of completely different dearer and time-consuming lab strategies,” says Júlia Domingo, co-first writer of the examine. “Our hope is that different scientists use the method to quickly and comprehensively map the allosteric websites of human proteins one after the other.”
One of many longer-term advantages of the method is its potential to check the perform and evolution of proteins. The authors of the examine consider that, if scaled up, the tactic may in the future end in advances that may exactly predict the properties of proteins from their amino acid sequences. If profitable, the authors argue this might usher in a brand new period of predictive molecular biology, permitting a lot sooner growth of latest drugs and clear, biology-based business.
“Whereas some instruments can predict a protein’s construction by studying its sequence, our methodology goes one step additional by telling us how a protein works. That is a part of an even bigger imaginative and prescient to make biology as engineerable as aeroplanes, bridges or computer systems. We've got confronted the identical challenges for over 70 years, nevertheless it seems they're extra tractable than we beforehand thought. If we succeed it would open a brand new subject with unprecedented potentialities,” concludes Dr. Lehner.
Reference: “Mapping the energetic and allosteric landscapes of protein binding domains” by Andre J. Faure, Júlia Domingo, Jörn M. Schmiedel, Cristina Hidalgo-Carcedo, Guillaume Diss and Ben Lehner, 6 April 2022, Nature.
DOI: 10.1038/s41586-022-04586-4
Post a Comment